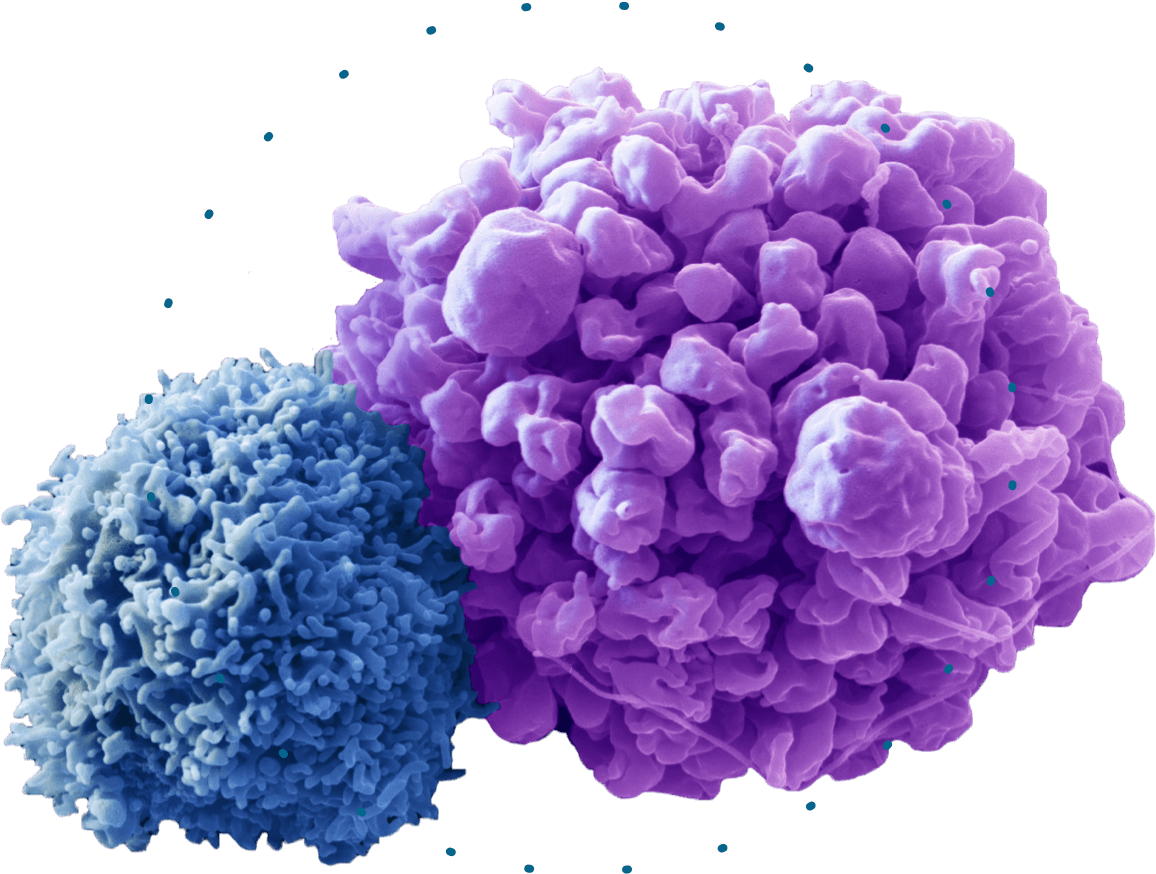
The Urbana site also has unfinished space available to further boost its capacity in the future, allowing employees to accommodate possible new scientific and technological advances in the cell therapy field, according to the release. Kite anticipates it will have 400 employees at its Urbana plant by the end of 2022, according to Tuesday's press release. Yescarta will be manufactured at the Urbana site. patients eligible for this treatment, Kite spokeswoman Tracy Rossin wrote in an email. Kite is a biopharmaceutical company engaged in the development of innovative cancer immunotherapies with a goal of providing rapid, long-term durable response a nd eliminating the burden of chronic care. The approval roughly doubled the number of U.S. Afterward, the cells are sent to the patient's hospital to be infused back into their body.ĬAR T-cell therapy is available at over 275 sites around the world, including more than 110 cancer hospitals in America, according to the release.Įarlier this month, the FDA approved Yescarta - Kite's commercially available CAR T-cell therapy - to be used for adult patients with large B-cell lymphoma who did not respond to their first treatment or relapsed within 12 months. We are driven by our mission to bring these. Today, we are a leader in engineered T cell therapy, which has changed the paradigm of cancer treatment as one of the biggest breakthroughs in medicine since the introduction of combination chemotherapy more than 60 years ago. The patient's T-cells, which function as part of their immune system, are then modified at Kite's manufacturing plants to recognize, attack and destroy their cancer cells. Kite has been at the forefront of cancer immunotherapy since 2009.
